From Wikipedia, the free encyclopedia
Homodihydrocapsaicin
Names
Preferred IUPAC name
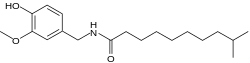
N -[(4-Hydroxy-3-methoxyphenyl)methyl]-9-methyldecanamide
Other names
Homodihydrocapsaicin N -Vanillyl-9-methyldecanamideVanillylamide of 9-methyldecanoic acid HDHC
Identifiers
ChemSpider
UNII
InChI=1S/C19H31NO3/c1-15(2)9-7-5-4-6-8-10-19(22)20-14-16-11-12-17(21)18(13-16)23-3/h11-13,15,21H,4-10,14H2,1-3H3,(H,20,22)
N Key: AKDLSISGGARWFP-UHFFFAOYSA-N
N InChI=1/C19H31NO3/c1-15(2)9-7-5-4-6-8-10-19(22)20-14-16-11-12-17(21)18(13-16)23-3/h11-13,15,21H,4-10,14H2,1-3H3,(H,20,22)
Key: AKDLSISGGARWFP-UHFFFAOYAS
O=C(NCc1cc(OC)c(O)cc1)CCCCCCCC(C)C
Properties
C 19 H 31 N O 3
Molar mass
−1
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
Chemical compound
Homodihydrocapsaicin is a capsaicinoid and analog and congener of capsaicin in chili peppers (Capsicum irritant . Homodihydrocapsaicin accounts for about 1% of the total capsaicinoids mixture[ 2] lipophilic colorless odorless crystalline to waxy compound. It produces "numbing burn" in the throat and is one of the most prolonged and difficult to rinse out. On the Scoville scale it has 8,600,000 SHU (Scoville heat units).[ 1]
^ a b Govindarajan, Sathyanarayana (1991). "Capsicum — Production, Technology, Chemistry, and Quality. Part V. Impact on Physiology, Pharmacology, Nutrition, and Metabolism; Structure, Pungency, Pain, and Desensitization Sequences". Critical Reviews in Food Science and Nutrition . 29 (6): 435–474. doi :10.1080/10408399109527536 . PMID 2039598 . ^ Bennett DJ, Kirby GW (1968). "Constitution and biosynthesis of capsaicin". J. Chem. Soc. C : 442. doi :10.1039/j39680000442 .
TRPA
Activators
4-Hydroxynonenal 4-Oxo-2-nonenal 4,5-EET 12S-HpETE 15-Deoxy-Δ12,14 -prostaglandin J2 α-Sanshool (ginger , Sichuan and melegueta peppers )Acrolein Allicin (garlic )Allyl isothiocyanate (mustard , radish , horseradish , wasabi )AM404 ASP-7663 Bradykinin Cannabichromene (cannabis )Cannabidiol (cannabis )Cannabigerol (cannabis )Cinnamaldehyde (cinnamon )CR gas (dibenzoxazepine; DBO) CS gas (2-chlorobenzal malononitrile) Cuminaldehyde (cumin )Curcumin (turmeric )Dehydroligustilide (celery )Diallyl disulfide Dicentrine (Lindera Farnesyl thiosalicylic acid Formalin Gingerols (ginger )Hepoxilin A3 Hepoxilin B3 Hydrogen peroxide Icilin Isothiocyanate JT-010 Ligustilide (celery , Angelica acutiloba Linalool (Sichuan pepper , thyme )Methylglyoxal Methyl salicylate (wintergreen )N-Methylmaleimide Nicotine (tobacco )Oleocanthal (olive oil )Paclitaxel (Pacific yew )Paracetamol (acetaminophen) PF-4840154 Phenacyl chloride Polygodial (Dorrigo pepper )Shogaols (ginger , Sichuan and melegueta peppers )Tear gases Tetrahydrocannabinol (cannabis )Tetrahydrocannabiorcol Thiopropanal S-oxide (onion )Umbellulone (Umbellularia californica )WIN 55,212-2 Blockers
TRPC
TRPM
TRPML
TRPP
TRPV
Activators
2-APB 5',6'-EET 9-HODE 9-oxoODE 12S-HETE 12S-HpETE 13-HODE 13-oxoODE 20-HETE α-Sanshool (ginger , Sichuan and melegueta peppers )Allicin (garlic )AM404 Anandamide Bisandrographolide (Andrographis paniculata Camphor (camphor laurel , rosemary , camphorweed , African blue basil , camphor basil )Cannabidiol (cannabis )Cannabidivarin (cannabis )Capsaicin (chili pepper )Carvacrol (oregano , thyme , pepperwort , wild bergamot , others)DHEA Diacyl glycerol Dihydrocapsaicin (chili pepper )Estradiol Eugenol (basil , clove )Evodiamine (Euodia ruticarpa Gingerols (ginger )GSK1016790A Heat Hepoxilin A3 Hepoxilin B3 Homocapsaicin (chili pepper )Homodihydrocapsaicin (chili pepper )Incensole (incense )Lysophosphatidic acid Low pH (acidic conditions)
Menthol (mint )N-Arachidonoyl dopamine N-Oleoyldopamine N-Oleoylethanolamide Nonivamide (PAVA) (PAVA spray )Nordihydrocapsaicin (chili pepper )Paclitaxel (Pacific yew )Paracetamol (acetaminophen) Phenylacetylrinvanil Phorbol esters (e.g., 4α-PDD )Piperine (black pepper , long pepper )Polygodial (Dorrigo pepper )Probenecid Protons RhTx Rutamarin (Ruta graveolens Resiniferatoxin (RTX) (Euphorbia resinifera /pooissonii Shogaols (ginger , Sichuan and melegueta peppers )Tetrahydrocannabivarin (cannabis )Thymol (thyme , oregano )Tinyatoxin (Euphorbia resinifera /pooissonii Tramadol Vanillin (vanilla )Zucapsaicin Blockers